| Metastasis | |
|---|---|
 Illustration showing hematogeneous metastasis | |
| Classification and external resources | |
| Specialty | Oncology |
| DiseasesDB | 28954 |
| MedlinePlus | 002260 |
Metastasis, or metastatic disease, is the spread of a cancer or other disease from one organ or part to another not directly connected with it. The new occurrences of disease thus generated are referred to as metastases /mə ˈtæs tə siːz/ (sometimes abbreviated "mets").[1][2] It was previously thought that only malignant tumor cells and infections have the capacity to metastasize(also spelled metastasise); new research has caused this to be reconsidered.[3] The existence of metastatic cancers in the absence of primary tumors also suggests that metastasis is not always caused by malignant cells that leave primary tumors.[4] Metastasis is a Greek word meaning "displacement", from μετά, meta, "next", and στάσις, stasis, "placement".
Cancer occurs after a single cell in a tissue is progressively genetically damaged to produce cells with uncontrolled proliferation. This uncontrolled proliferation by mitosis produces aprimary heterogeneic tumour. The cells which constitute the tumor eventually undergo metaplasia, followed by dysplasia then anaplasia, resulting in a malignant phenotype. This malignancy allows for invasion into the circulation, followed by invasion to a second site for tumorigenesis.
Some cancer cells acquire the ability to penetrate the walls of lymphatic and/or blood vessels, after which they are able to circulate through the bloodstream (circulating tumor cells) to other sites and tissues in the body. This process is known (respectively) as lymphatic or hematogenous spread.
After the tumor cells come to rest at another site, they re-penetrate the vessel or walls and continue to multiply, eventually forming another clinically detectable tumor. This new tumor is known as a metastatic (or secondary) tumor. Metastasis is one of three hallmarks of malignancy (contrast benign tumors).[5] Most neoplasms can metastasize, although in varying degrees (e.g.,basal cell carcinoma rarely metastasize).[5]
When tumor cells metastasize, the new tumor is called a secondary or metastatic tumor, and its cells are similar to those in the original tumor. This means, for example, that, if breast cancermetastasizes to the lungs, the secondary tumor is made up of abnormal breast cells, not of abnormal lung cells. The tumor in the lung is then called metastatic breast cancer, not lung cancer.
Initially, nearby lymph nodes are struck early.[6] The lungs, liver, brain, and bones are the most common metastasis locations from solid tumors.[6]
Although advanced cancer may cause pain, it is often not the first symptom.
Some patients, however, do not show any symptoms.[6] When the organ gets a metastatic disease it begins to shrink until its lymph nodes burst, or undergo lysis.
Pathophysiology[edit]Metastatic tumors are very common in the late stages of cancer. The spread of metastasis may occur via the blood or the lymphatics or through both routes. The most common places for the metastases to occur are the lungs, liver,brain, and the bones.[7]
Factors involved[edit]Metastasis is a complex series of steps in which cancer cells leave the original tumor site and migrate to other parts of the body via the bloodstream, the lymphatic system, or by direct extension. To do so, malignant cells break away from the primary tumor and attach to and degrade proteins that make up the surrounding extracellular matrix (ECM), which separates the tumor from adjoining tissues. By degrading these proteins, cancer cells are able to breach the ECM and escape. The location of the metastases is not always random, with different types of cancer tending to spread to particular organs and tissues at a rate that is higher than expected by statistical chance alone.[8] Breast cancer, for example, tends to metastasize to the bones and lungs. This specificity seems to be mediated by soluble signal molecules such as chemokines[9] and transforming growth factor beta.[10] The body resists metastasis by a variety of mechanisms through the actions of a class of proteins known as metastasis suppressors, of which about a dozen are known.[11]
Human cells exhibit three kinds of motion: collective motility, mesenchymal-type movement, and amoeboid movement. Cancer cells often opportunistically switch between different kinds of motion. Some cancer researchers hope to find treatments that can stop or at least slow down the spread of cancer by somehow blocking some necessary step in one or the other or both kinds of motion.[12]
Cancer researchers studying the conditions necessary for cancer metastasis have discovered that one of the critical events required is the growth of a new network of blood vessels, called tumor angiogenesis.[13] It has been found thatangiogenesis inhibitors would therefore prevent the growth of metastases.[5]
Several different cell types are critical to tumor growth. In particular, endothelial progenitor cells have been shown to have a strong influence on the growth of tumor blood vessels. This finding was published in the journalsScience (2008) and Genes and Development (2007) together with the fact that endothelial progenitor cells are critical for metastasis and angiogenesis.[14][15] The importance of endothelial progenitor cells in tumor growth, angiogenesis and metastasis has been confirmed by a recent publication in Cancer Research (August 2010). This seminal paper has demonstrated that endothelial progenitor cells can be marked using the Inhibitor of DNA Binding 1 (ID1). This novel finding meant that investigators were able to track endothelial progenitor cells from the bone marrow to the blood to the tumor-stroma and even incorporated in tumor vasculature. This finding of endothelial progenitor cells incorporated in tumor vasculature proves the importance of this cell type in blood vessel development in a tumor setting and metastasis. Furthermore, ablation of the endothelial progenitor cells in the bone marrow lead to a significant decrease in tumor growth and vasculature development. Therefore, endothelial progenitor cells are very important in tumor biology and present novel therapeutic targets.[16]
NFAT transcription factors are implicated in breast cancer, more specifically in the process of cell motility at the basis of metastasis formation. Indeed, NFAT1 (NFATC2) and NFAT5 are pro-invasive and pro-migratory in breast carcinoma[17][18] and NFAT3 (NFATc4) is an inhibitor of cell motility.[19] NFAT1 regulates the expression of the TWEAKR and its ligand TWEAK with the Lipocalin 2 to increase breast cancer cell invasion [20] and NFAT3 inhibits Lipocalin 2 expression to blunt the cell invasion.[19]
Epigenetic regulation also plays an important role in the metastatic outgrowth of disseminated tumor cells. Metastases display alterations in histone modifications, such as H3K4-methylation and H3K9-methylation, when compared to matching primary tumors.[21] These epigenetic modifications in metastases may allow the proliferation and survival of disseminated tumor cells in distant organs.[22]
Routes of metastasis[edit]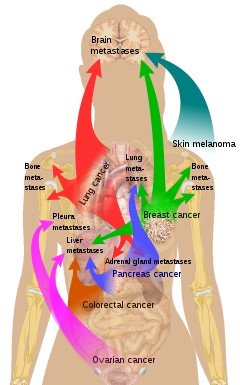
Metastasis occurs by following four routes:
1. Transcoelomic[edit]The spread of a malignancy into body cavities can occur via penetrating the surface of the peritoneal, pleural, pericardial, or subarachnoid spaces. For example, ovarian tumors can spread transperitoneally to the surface of the liver.
2. Lymphatic spread[edit]Lymphatic spread allows the transport of tumor cells to lymph nodes and ultimately, to other parts of the body. This is the most common route of metastasis for carcinomas. In contrast, it is uncommon for a sarcoma to metastasize via this route. It is worth noting that the lymphatic system does eventually drain into the systemic venous system via the azygous vein, and therefore these metastatic cells can eventually spread through the haematogenous route.
This is typical route of metastasis for sarcomas, but it is also the favored route for certain types of carcinoma, such as those originating in the kidney (renal cell carcinoma). Because of their thinner walls, veins are more frequently invaded than are arteries, and metastasis tends to follow the pattern of venous flow.
4. Transplantation or implantation[edit]Cancer cells may spread to lymph nodes (regional lymph nodes) near the primary tumor. This is called nodal involvement, positive nodes, or regional disease. ("Positive nodes" is a term that would be used by medical specialists to describe a patient's condition, meaning that the patient's lymph nodes near the primary tumor tested positive for malignancy. It is common medical practice to test by biopsy at least two lymph nodes near a tumor site when doing surgery to examine or remove a tumor.) Localized spread to regional lymph nodes near the primary tumor is not normally counted as metastasis, although this is a sign of worse prognosis. Transport through lymphatics is the most common pathway for the initial dissemination of carcinomas.[5]
Organ-specific targets[edit]There is a propensity for certain tumors to seed in particular organs. This was first discussed as the "seed and soil" theory by Stephen Paget over a century ago, in 1889. The propensity for a metastatic cell to spread to a particular organ is termed 'organotropism'. For example, prostate cancer usually metastasizes to the bones. In a similar manner, colon cancer has a tendency to metastasize to the liver. Stomach cancer often metastasises to the ovary in women, then it is called a Krukenberg tumor.
According to the "seed and soil" theory, it is difficult for cancer cells to survive outside their region of origin, so in order to metastasize they must find a location with similar characteristics.[24] For example, breast tumor cells, which gather calcium ions from breast milk, metastasize to bone tissue, where they can gather calcium ions from bone. Malignant melanoma spreads to the brain, presumably because neural tissue and melanocytes arise from the same cell line in the embryo.[25]
In 1928, James Ewing challenged the "seed and soil" theory and proposed that metastasis occurs purely by anatomic and mechanical routes. This hypothesis has been recently utilized to suggest several hypotheses about the life cycle of circulating tumor cells (CTCs) and to postulate that the patterns of spread could be better understood through a 'filter and flow' perspective.[26]
Metastasis and primary cancer[edit]It is theorized that metastasis always coincides with a primary cancer, and, as such, is a tumor that started from a cancer cell or cells in another part of the body. However, over 10% of patients presenting to oncology units will have metastases without a primary tumor found. In these cases, doctors refer to the primary tumor as "unknown" or "occult," and the patient is said to have cancer of unknown primary origin (CUP) or unknown primary tumors (UPT).[27] It is estimated that 3% of all cancers are of unknown primary origin.[28] Studies have shown that, if simple questioning does not reveal the cancer's source (coughing up blood—"probably lung", urinating blood—"probably bladder"), complex imaging will not either.[28] In some of these cases a primary tumor may appear later.
The use of immunohistochemistry has permitted pathologists to give an identity to many of these metastases. However, imaging of the indicated area only occasionally reveals a primary. In rare cases (e.g., of melanoma), no primary tumor is found, even on autopsy. It is therefore thought that some primary tumors can regress completely, but leave their metastases behind.
Common sites of origin[edit]The cells in a metastatic tumor resemble those in the primary tumor. Once the cancerous tissue is examined under a microscope to determine the cell type, a doctor can usually tell whether that type of cell is normally found in the part of the body from which the tissue sample was taken.
For instance, breast cancer cells look the same whether they are found in the breast or have spread to another part of the body. So, if a tissue sample taken from a tumor in the lung contains cells that look like breast cells, the doctor determines that the lung tumor is a secondary tumor. Still, the determination of the primary tumor can often be very difficult, and the pathologist may have to use several adjuvant techniques, such as immunohistochemistry, FISH (fluorescent in situ hybridization), and others. Despite the use of techniques, in some cases the primary tumor remains unidentified.
Metastatic cancers may be found at the same time as the primary tumor, or months or years later. When a second tumor is found in a patient that has been treated for cancer in the past, it is more often a metastasis than another primary tumor.
It was previously thought that most cancer cells have a low metastatic potential and that there are rare cells that develop the ability to metastasize through the development of somatic mutations.[30] According to this theory, diagnosis of metastatic cancers is only possible after the event of metastasis. Traditional means of diagnosing cancer (e.g. a biopsy) would only investigate a subpopulation of the cancer cells and would very likely not sample from the subpopulation with metastatic potential.[31]
The somatic mutation theory of metastasis development has not been substantiated in human cancers. Rather, it seems that the genetic state of the primary tumor reflects the ability of that cancer to metastasize.[31] Research comparing gene expression between primary and metastatic adenocarcinomas identified a subset of genes whose expression could distinguish primary tumors from metastatic tumors, dubbed a "metastatic signature."[31] Up-regulated genes in the signature include: SNRPF, HNRPAB, DHPS and securin. Actin, myosin and MHC class II down-regulation was also associated with the signature. Additionally, the metastatic-associated expression of these genes was also observed in some primary tumors, indicating that cells with the potential to metastasize could be identified concurrently with diagnosis of the primary tumor.[32]
Expression of this metastatic signature has been correlated with a poor prognosis and has been shown to be consistent in several types of cancer. Prognosis was shown to be worse for individuals whose primary tumors expressed the metastatic signature.[31] Additionally, the expression of these metastatic-associated genes was shown to apply to other cancer types in addition to adenocarcinoma. Metastases of breast cancer, medulloblastoma and prostate cancer all had similar expression patterns of these metastasis-associated genes.[31]
The identification of this metastasis-associated signature provides promise for identifying cells with metastatic potential within the primary tumor and hope for improving the prognosis of these metastatic-associated cancers. Additionally, by identifying the genes whose expression is changed in metastasis offers potential targets to inhibit metastasis.[31]
Micrograph of thyroid cancer (papillary thyroid carcinoma) in a lymph node of the neck. H&E stain
CT image of multiple liver metastases
CT image of a lung metastasis
Metastasis proven byliver biopsy (tumor (adenocarcinoma) - lower two-thirds of image). H&E stain.
Treatment and survival is determined, to a great extent, by whether or not a cancer remains localized or spreads to other locations in the body. If the cancer metastasizes to other tissues or organs it usually dramatically increases a patient's likelihood of death (i.e. the "prognosis"). Some cancers - such as some forms of leukemia, a cancer of the blood, or malignancies in the brain - can kill without spreading at all.
Once a cancer has metastasized it may still be treated with radiosurgery, chemotherapy, radiation therapy, biological therapy, hormone therapy, surgery, or a combination of these interventions ("multimodal therapy"). The choice of treatment depends on a large number of factors, including the type of primary cancer, the size and location of the metastases, the patient's age and general health, and the types of treatments used previously. In patients diagnosed with CUP it is often still possible to treat the disease even when the primary tumor cannot be located.
Current treatments are rarely able to cure metastatic cancer though some tumors, such as testicular cancer and thyroid cancer, are usually still curable.
History[edit]In March 2014 researchers discovered the oldest complete example of a human with metastatic cancer. The tumors had developed in a 3,000-year-old skeleton found in 2013 in a tomb in Sudan dating back to 1200 BC. The skeleton was analyzed using radiography and a scanning electron microscope. These findings were published in the Public Library of Science journal.[33][34][35]
See also[edit]Archiver|干细胞之家 ( 吉ICP备2021004615号-3 )
GMT+8, 2026-2-5 11:27
Powered by Discuz! X1.5
© 2001-2010 Comsenz Inc.